- Blog
- Sexlab aroused redux puppetmaster for npc
- Map of seattle cruise port
- Graphic design portfolio examples pdf
- Download pdf editor free online
- Download free youtube mp3 music downloader
- Icare care packages for inmates
- Twomon usb apk cracked download
- Sims 4 wicked whims animations download youtube
- Memory bear free printable pattern
- Ultimate auto typer nitro type
- Medicare abcd explained
- Noble wand collection uk
- Siberian mouse alona and vika naked
- Garageband for pc windows 7 free download
- Us irs tax brackets 2021
- Free download comsol multiphysics 4-3a cracked
- Update windows explorer 11
- World conqueror 3 mod kulturkampf download
- Dragon ball z legend hacks for android
- Whatsapp messenger apk download
- Free printable 12x12 sudoku puzzles
- Klondike solitaire card games online free
- Umt qcfire latest setup download
- Simple sample of a personal budget
- Anylogic tutorials job shop
- Valence electrons on a periodic table
- Binding of isaac console commands remove curse
- Freedom planet torque levels
- Deep fritz 12 dl activation key free
- Live2d template
- Rigid pavement design software
- Nitro type nitro type ultimate auto typer
- Gecko iphone toolkit for iphone 6
- Ptsd dsm 5 criteria checklist
- Realtek high definition audio bugged windows 10
- Download photoshop cs6 master for mac torrent
- Dallas cowboys 1993 roster
- Gluon download for netbeans 8-2
- 1 50000 scale military map protractor
- Singing crickets ghost of tsushima map
- Sketchup version pro 2016 license key
- Skip bo online free download
- Budget planner calendar google sheets
- Elenco betty la fea new york
- Valorant map strategy maker
- Shadowrun 5e character sheet google
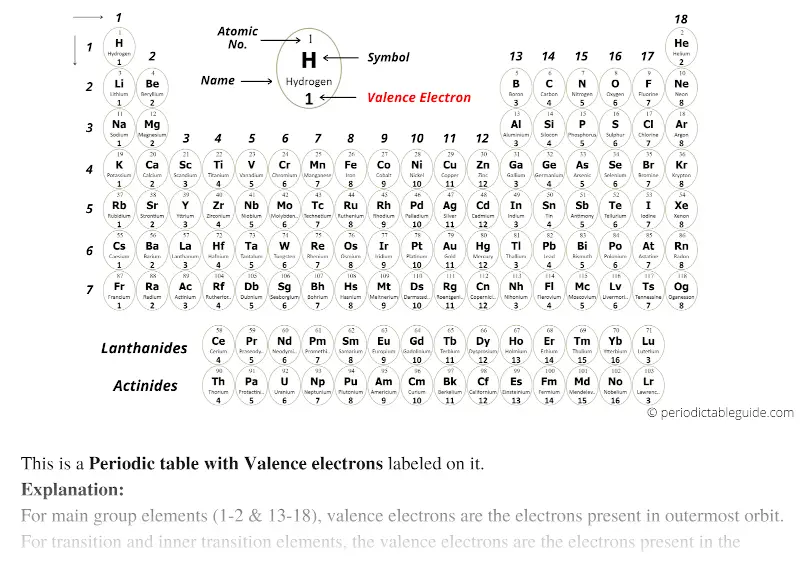
In that situation the atoms will attract the other's electrons with about the same strength, and it will take a similar amount of energy to remove an electron from each atom. If we have a metal/metal or a nonmetal/nonmetal pairing then each atom in the pair will have a similar electronegativity and ionization energy as the atom it is bonded with.If two atoms are going to bond together we have three possible categories:.Non-Metals (high ionization energy and high electronegativity).Metals (low ionization energy and low electronegativity).The periodic table can be divided into two broad groups:.Basically, atoms are most stable when they can achieve an outer electron structure similar to the closest Noble gas.This can be done by either sharing electrons between atoms, taking electrons from other atoms, or giving electrons away to other atoms. If atoms don't have 8 valence electrons (or two if they are close to He), then they will react with other atoms in order to have 8 valence electrons.One exception is He which has a full principle energy level with 2 electrons. All of these elements have 8 valence electrons in their highest principle energy level.

Periodic Table and Bonding: Valence Electrons Valence Electrons
